Blog
Foundational Biomarkers to Assess Health: What They Mean and What to Look For

Why Foundational Biomarkers Matter for Preventive Health
Most people only get lab work done when something is already wrong. A symptom appears, prompts a visit, and the testing that follows is aimed at confirming a diagnosis rather than catching a problem before it develops into one. That reactive model works for acute illness. It works poorly for the chronic conditions — cardiovascular disease, type 2 diabetes, hormonal dysregulation, neurodegeneration — that develop over years or decades before producing symptoms that anyone notices.
Foundational biomarkers for health are the measurable indicators that show what’s happening at the cellular, metabolic, hormonal, and inflammatory levels before symptoms appear. They’re the data layer that transforms a vague sense of ‘feeling off’ or ‘not as sharp as I used to be’ into specific, actionable findings. And they’re the objective baseline against which any health intervention can be measured to determine whether it’s actually working. Understanding your lab work in depth starts with knowing which markers tell you the most and why.
The goal of comprehensive biomarker testing at LIVV Natural is not to screen for disease — it’s to identify the specific biological imbalances that are driving a person toward disease risk or suboptimal function, and to use that information to build a treatment protocol that addresses those mechanisms directly. This is a different approach from standard annual physicals, which typically screen for conditions that already exist.
What Foundational Biomarkers Are — and What They Measure
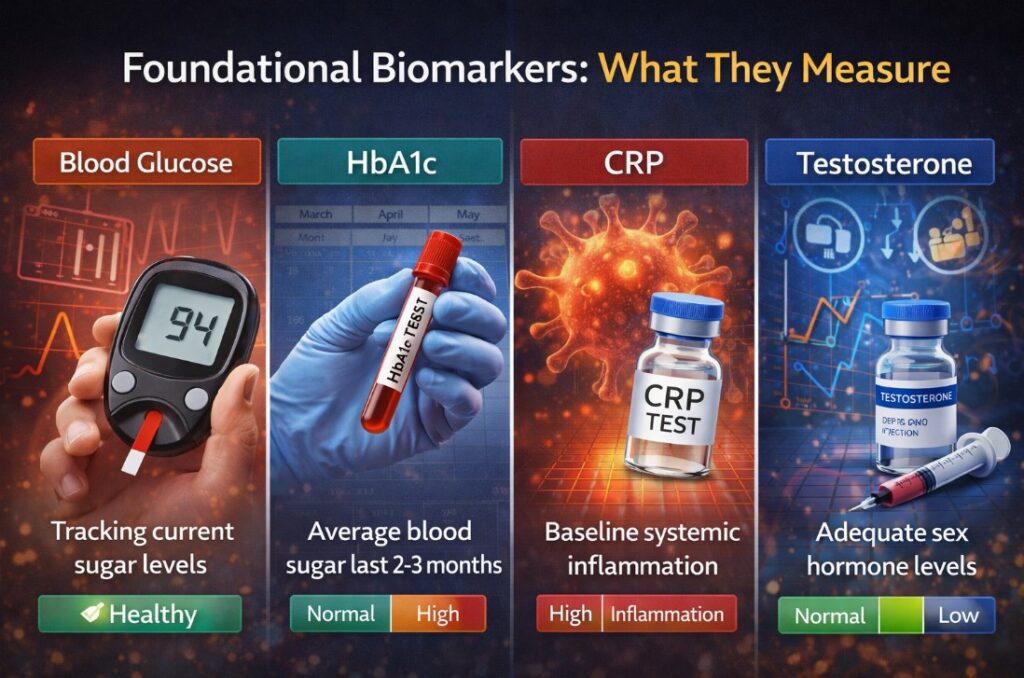
A biomarker is any measurable biological characteristic that indicates what’s happening in the body. Blood glucose tells you how your cells are processing sugar right now. HbA1c tells you what your average blood sugar has been over the past two to three months. CRP tells you how much systemic inflammation is running at baseline. Testosterone levels tell you whether your endocrine system is producing adequate sex hormones for your age and health profile. Each of these is a biomarker — a measurable data point that reflects a specific biological process.
The word ‘foundational’ in the context of biomarker panels refers to markers that are informative across a broad range of health concerns — not niche markers useful only in specific disease contexts, but the kind of markers that tell you something actionable regardless of your age, health history, or chief complaint. Foundational biomarker panels cover metabolism, inflammation, hormones, cognitive health, and longevity markers because these systems interact with each other and collectively determine how well the body is functioning and how quickly it’s aging.
What makes biomarker testing particularly useful is the gap it reveals between subjective experience and objective biology. Many people feel fine at CRP levels that predict cardiovascular events years later. Many people have hormonal imbalances for years before those imbalances produce symptoms obvious enough to seek medical attention. Biomarkers close that gap — they give you a view of what’s happening inside the body before the body signals distress through symptoms you can’t ignore.
The Four Types of Biomarkers and What Each Does
Not all biomarkers serve the same clinical function. Understanding the different categories helps clarify what a panel is telling you and what to do with the information.
Diagnostic biomarkers confirm the presence of an existing condition. A fasting glucose above 126 mg/dL on two separate tests is diagnostic for diabetes. TSH above a certain threshold is diagnostic for hypothyroidism. These markers are used to identify what’s already there — they don’t predict; they confirm.
Predictive biomarkers indicate future risk before disease onset. APOE genotype is the clearest example: carrying the APOE ε4 allele significantly increases Alzheimer’s disease risk without causing it. Elevated hs-CRP in an otherwise healthy person predicts future cardiovascular events. Telomere length below age-matched norms predicts accelerated cellular aging and earlier onset of age-related disease. Predictive biomarkers are where preventive medicine does its most useful work — they allow intervention before the process becomes pathological.
Prognostic biomarkers predict how a disease will progress. IGF-1 levels in cancer, HbA1c trajectory in diabetes, or cortisol patterns in adrenal insufficiency all help forecast the likely course of a condition already identified. In a longevity context, prognostic markers help track whether biological aging is accelerating or decelerating over time in response to interventions.
Monitoring biomarkers assesses whether a treatment is working. If you start a peptide protocol to support GH output, follow-up IGF-1 levels tell you whether the protocol is producing the expected hormonal response. If you begin an anti-inflammatory dietary intervention, CRP retesting at 8 to 12 weeks tells you whether systemic inflammation has dropped. Monitoring biomarkers is the feedback mechanism that makes personalized medicine genuinely personalized — they close the loop between intervention and outcome.
Metabolic Health Biomarkers
Metabolic dysfunction is one of the most common and underdiagnosed contributors to accelerated aging, low energy, cognitive decline, and cardiovascular risk. The foundational metabolic markers tell you where your body is on the spectrum from optimal insulin sensitivity to metabolic syndrome — and they reveal this picture years before any single marker crosses a diagnostic threshold.
Fasting glucose and fasting insulin: These two markers together tell you far more than either does alone. Fasting glucose reflects how well cells are clearing glucose from the bloodstream. But insulin can be elevated long before glucose rises — for years, in some cases — as the pancreas compensates for growing cellular insulin resistance by producing more insulin to achieve the same effect. The HOMA-IR calculation (fasting glucose × fasting insulin ÷ 405) quantifies insulin resistance from these two numbers and is one of the most actionable early indicators in metabolic health testing.
Hemoglobin A1c (HbA1c): HbA1c reflects average blood glucose over the preceding two to three months by measuring the percentage of hemoglobin that has glucose attached to it. It doesn’t require fasting, is not affected by a single high-carbohydrate meal, and provides a more stable picture of glycemic exposure than a single fasting glucose measurement. A biomarker for prediabetes and diabetes identifies HbA1c as the most commonly used clinical marker for diagnosing dysglycemia, though it has limitations in certain populations, which is why the LIVV panel combines it with fasting insulin and glucose for a more complete metabolic picture.
Lipid panel: Standard lipid panels report total cholesterol, LDL, HDL, and triglycerides. A more complete picture also includes LDL particle size and number — small, dense LDL particles are significantly more atherogenic than large, buoyant LDL at the same total LDL concentration, which is why LDL particle testing changes clinical risk assessments in a meaningful percentage of patients who look acceptable on a standard panel.
Inflammation Biomarkers: CRP, IL-6, and Why Chronic Inflammation Matters
Chronic low-grade inflammation is one of the strongest and most consistent predictors of accelerated biological aging and age-related disease. It is measurable, modifiable, and often entirely asymptomatic for years or decades before its downstream effects — cardiovascular disease, cognitive decline, cancer, metabolic syndrome — become clinically apparent.
High-sensitivity CRP (hs-CRP) is the most widely used inflammation biomarker in preventive medicine. A 2022 meta-analysis in the International Journal of Molecular Sciences found that elevated hs-CRP was associated with a hazard ratio of 1.19 (95% CI: 1.09–1.30) for predicting cardiovascular events in asymptomatic middle-aged adults — making it the strongest single biomarker identified across 25 included studies for primary CVD risk prediction in people without prior cardiovascular history. The conventional risk cut-points are: below 1 mg/L (low risk), 1–3 mg/L (intermediate risk), above 3 mg/L (high risk), with a caveat that acute infection or injury can transiently elevate CRP independent of chronic inflammatory burden.
Interleukin-6 (IL-6): IL-6 is the upstream cytokine that drives CRP production in the liver. Testing both IL-6 and CRP together gives a more complete picture of inflammatory signaling — IL-6 is particularly relevant in the context of visceral fat (which secretes IL-6 independently of other inflammatory triggers) and stress-related immune dysregulation. Elevated IL-6 is also strongly associated with sarcopenia, cognitive decline, and cardiovascular risk independent of CRP.
Homocysteine: Homocysteine is an amino acid that accumulates when B vitamin status (particularly B12, B6, and folate) is insufficient or when methylation pathways are impaired. Elevated homocysteine independently predicts cardiovascular disease, stroke, and cognitive decline — including Alzheimer’s disease risk. It’s also one of the more easily correctable markers through targeted B vitamin supplementation, which makes it both diagnostically and therapeutically useful in a biomarker panel.
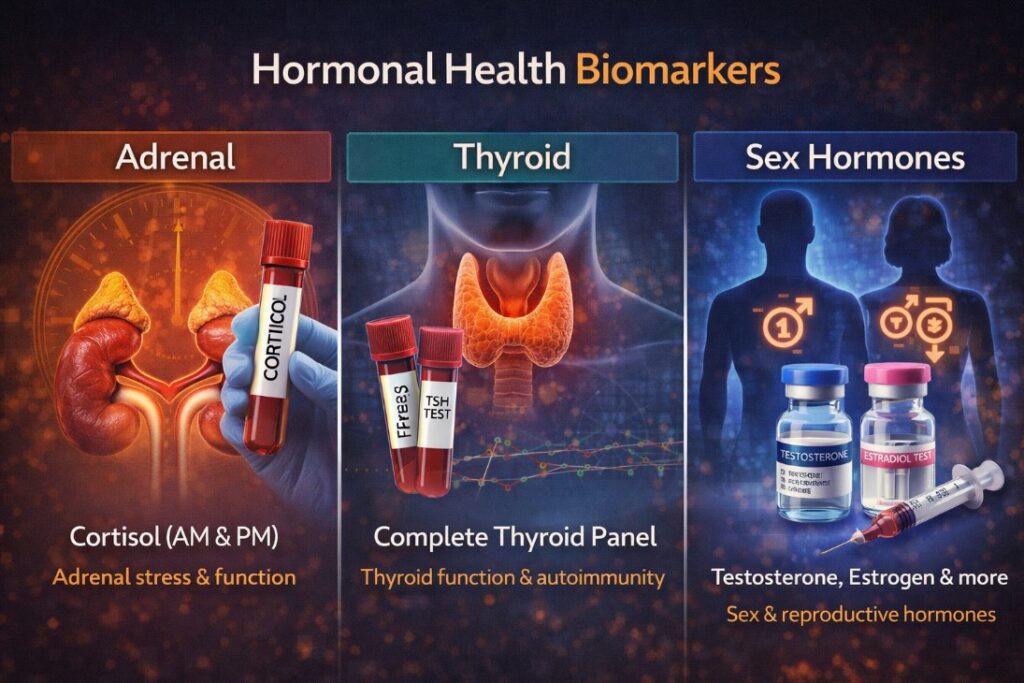
Hormonal Health Biomarkers
Hormonal balance affects almost every aspect of health — energy, body composition, mood, sleep, cognitive function, libido, and the rate of biological aging. The hormonal biomarker panel at LIVV covers the major axes: adrenal, thyroid, and sex hormones.
Cortisol (AM and PM): Cortisol follows a diurnal rhythm — high in the morning, declining through the day. AM cortisol should be at its peak; PM cortisol should be substantially lower. Disruptions to this pattern — chronically elevated baseline, flat diurnal curve, or early-morning low that fails to peak — each indicate different types of HPA axis dysregulation with different clinical implications. The cortisol testing and adrenal health guide on the LIVV blog covers the interpretation of cortisol patterns in detail.
Thyroid panel (TSH, Free T3, Free T4, Reverse T3, antibodies): Standard thyroid testing typically includes only TSH, which is a pituitary signal that reflects whether the brain thinks thyroid hormone levels are adequate — not whether the thyroid is actually producing usable hormone, not whether T4 is being converted to the active T3 form, and not whether autoimmune activity (Hashimoto’s) is present. A complete thyroid panel adds Free T3 and Free T4 (the usable forms), Reverse T3 (which blocks T3 receptors when elevated), and thyroid antibodies (TPO and TG). Many people with normal TSH have abnormal Free T3/T4 ratios, elevated Reverse T3, or positive antibodies — all of which explain symptoms that TSH alone misses. Thyroid health and its effect on overall function depend on this full picture.
Sex hormones (testosterone, estrogen, progesterone, DHEA-S, SHBG): For both men and women, total testosterone alone is an incomplete measure. Free testosterone — the bioavailable fraction not bound to sex hormone-binding globulin — is what actually drives tissue effects. High SHBG can produce normal total testosterone with clinically low free testosterone. Estradiol in men and progesterone in women are frequently omitted from standard panels despite their direct effects on mood, sleep, bone density, and cardiovascular health. DHEA-S provides a window into adrenal androgen reserve and tends to decline predictably with age. The full sex hormone panel from men’s hormone optimization and women’s hormone optimization at LIVV starts from a complete picture rather than isolated values.
IGF-1: IGF-1 is the primary mediator of growth hormone effects in peripheral tissue. Because GH itself is secreted in pulses and has a short half-life, a single GH measurement is largely meaningless. IGF-1 provides a stable, integrated measure of GH activity over days and is used to monitor the effectiveness of GH-stimulating peptide protocols.
Cognitive Health Biomarkers
Most cognitive decline is not sudden or genetic — it develops over years through accumulating biological changes that are measurable long before the cognitive effects become apparent. The cognitive health biomarker category covers the markers most predictive of neurological aging and dementia risk.
Brain-derived neurotrophic factor (BDNF): BDNF is the primary growth factor for neurons — it supports the survival, growth, and maintenance of nerve cells, and is the molecular mediator of many of the cognitive benefits of exercise, sleep, and stress management. BDNF levels decline with age and are lower in people with cognitive impairment and Alzheimer’s disease. A study in Scientific Reports examining community-dwelling older adults found that older age, less physical activity, hippocampal atrophy, and lower serum BDNF levels all independently predicted memory impairment, with BDNF appearing as a significant factor after controlling for the other variables. Measuring BDNF gives a direct read on neuroplasticity reserve.
APOE genotype: APOE is a gene with three common variants (ε2, ε3, ε4). Carrying one copy of the ε4 allele raises lifetime Alzheimer’s risk approximately 3-fold; two copies raises it 8–12 fold. This doesn’t determine fate — many ε4 carriers never develop Alzheimer’s — but knowing your APOE status informs the urgency of protective lifestyle interventions (sleep optimization, inflammation reduction, aerobic exercise, DHA intake) and helps prioritize which cognitive markers to monitor most closely over time.
Homocysteine and B vitamin status: Homocysteine is both an inflammation marker and a cognitive health marker. Its association with cognitive decline and dementia is independent of its cardiovascular effects. B12 and folate deficiency are among the most common correctable contributors to elevated homocysteine, and B vitamin status is under-assessed in most standard panels despite being straightforward to measure and straightforward to address.
Oxidative stress markers: Oxidative damage to neurons accumulates over decades and contributes to the structural changes underlying neurodegenerative disease. Markers like 8-OHdG (a DNA oxidation product) and F2-isoprostanes (lipid oxidation products) provide a direct measure of oxidative burden on cells. These markers also guide antioxidant therapy — glutathione IV protocols, for instance, are more precisely dosed when baseline oxidative stress is measured rather than assumed.
Longevity Biomarkers: Telomeres, Mitochondria, NAD+, and AGEs
Longevity biomarkers go beyond disease risk and into the underlying biology of aging itself — measuring how fast cells are aging, how well cellular maintenance is running, and where the biological age gaps are that accumulate into chronic disease risk over time.
Telomere length: Telomeres are the protective caps on chromosomes that shorten with each cell division. Critically short telomeres trigger cellular senescence — the state where cells stop dividing but remain metabolically active in ways that accelerate surrounding tissue aging. Telomere length provides a cumulative measure of cellular replicative history and stress exposure, and correlates with biological age vs. chronological age discrepancies. People with shorter telomeres relative to their age-matched population show higher risks of cardiovascular disease, cognitive decline, and early mortality.
Mitochondrial function: Mitochondrial dysfunction is both a cause and consequence of cellular aging. As mitochondria decline in efficiency, they produce more reactive oxygen species relative to ATP output, creating an oxidative environment that accelerates DNA damage and senescence. Assessing mitochondrial health through indirect markers — CoQ10 levels, lactate-to-pyruvate ratios, organic acid profiles — identifies mitochondrial decline before it produces obvious symptoms like fatigue or exercise intolerance.
NAD+ levels: NAD+ IV therapy is most precisely applied when baseline NAD+ depletion is confirmed. NAD+ declines 40 to 50% between early adulthood and middle age across most tissues, impairing the sirtuin and PARP enzyme systems that govern DNA repair, mitochondrial biogenesis, and cellular senescence. Measuring NAD+ precursors and relevant metabolites gives an objective indicator of cellular energy status that supplements the clinical picture from other longevity markers.
Advanced glycation end products (AGEs): AGEs form when glucose binds non-enzymatically to proteins or lipids, accumulating in long-lived tissues like arterial walls, kidney tissue, and collagen. Skin autofluorescence — a non-invasive measurement of AGE accumulation in skin tissue — correlates with AGE burden across other tissues and is predictive of cardiovascular risk and diabetic complications independent of HbA1c.
Why Biomarker Assessment Changes How You Approach Health
The most direct benefit of comprehensive biomarker testing is that it converts health management from reactive to proactive. Standard medicine identifies problems after they develop symptoms. Biomarker testing identifies the upstream biological conditions that produce those symptoms, years before they manifest clinically. Someone who discovers at 45 that they have a CRP of 4 mg/L, a pre-diabetic HbA1c of 5.9%, and low free testosterone is not sick by any clinical diagnostic standard. But they are in a biological trajectory toward cardiovascular disease, metabolic syndrome, and hormonal decline. Acting on that information at 45 produces radically different outcomes than acting at 55 after the conditions have advanced.
The second benefit is that biomarker data makes interventions accountable. Diet changes, sleep improvements, supplements, IV protocols, peptide therapy, hormone optimization — all of these produce biological effects that show up in lab values. Testing before and after an intervention at 8 to 12 weeks tells you whether what you’re doing is actually moving the numbers in the right direction. Without that feedback, you’re operating on an assumption.
The third benefit is personalization. A comprehensive blood panel reveals that two people with nearly identical symptoms — fatigue, brain fog, difficulty maintaining body weight — can have completely different underlying drivers: one might have elevated IL-6 and insulin resistance driving their symptoms; another might have low Free T3 and low BDNF. The same treatment protocol addresses neither person optimally. Starting from individual biomarker data is what makes a protocol genuinely personalized rather than standardized.
How Biomarkers Are Tested
Blood testing remains the most comprehensive and widely validated method for most foundational biomarkers. A single blood draw at LIVV covers metabolic markers, inflammatory indicators, hormones, lipids, micronutrients, and longevity markers — the full picture from one collection. Fasting for 10 to 12 hours beforehand improves accuracy for glucose, insulin, lipids, and some hormone panels.
Saliva testing is particularly useful for cortisol measurement because it allows multiple collections across the day (morning, noon, afternoon, evening) without blood draws, which are themselves a stressor that can transiently affect cortisol levels. Saliva sex hormone testing is also available and can be useful in specific clinical contexts where the free fraction of hormones is more relevant than total serum levels.
At-home versus in-clinic testing has tradeoffs covered in more detail in the at-home vs. in-clinic testing comparison on the LIVV blog. The short version: in-clinic testing under consistent, standardized conditions produces more reliable results for most markers, particularly those where sample handling or timing affects values. At-home testing has improved significantly for a subset of markers and is reasonable for monitoring when in-clinic visits are infrequent.
How Often to Test
A baseline comprehensive panel is the starting point — you can’t track change without a baseline. For people actively running health optimization protocols, retesting at 8 to 12 weeks is standard for markers that respond quickly to intervention (CRP, insulin, cortisol, thyroid). Quarterly testing is appropriate for active protocols where regular adjustments are expected. Twice-yearly testing is the minimum for maintenance monitoring — sufficient to catch meaningful changes without over-testing markers that respond slowly.
Certain markers — APOE genotype, for instance — don’t need retesting because they don’t change. Telomere length and epigenetic age testing are most informative when tested every 6 to 12 months because changes accumulate slowly and are difficult to detect at shorter intervals. Mitochondrial function markers and NAD+ levels respond to IV protocols and lifestyle changes over weeks to months, making quarterly retesting reasonable when those interventions are active.
The principle across all these timeframes is that testing without action is wasteful, and action without testing is guesswork. The monitoring schedule should be calibrated to the protocols in use — frequent enough to catch whether something isn’t working before too much time passes, infrequent enough that meaningful change has had time to accumulate between measurements.
LIVV Natural’s Biomarker-Driven Approach to Personalized Health
LIVV Natural’s naturopathic medicine practice in San Diego builds every treatment protocol from a comprehensive biomarker baseline. The intake process begins with a full panel of metabolic, inflammatory, hormonal, cognitive, and longevity markers — interpreted by the LIVV medical team to identify which biological pathways are most in need of support and what interventions are most likely to move those specific numbers.
What distinguishes this approach from standard primary care is the scope of testing and the treatment philosophy. Standard annual physicals include a subset of the markers covered in a comprehensive LIVV panel, interpreted primarily through a diagnostic lens — looking for conditions that already qualify for treatment. LIVV’s interpretation starts from the question of what’s driving a patient’s biological trajectory, which means identifying patterns that don’t yet meet diagnostic criteria but are clearly heading somewhere unfavorable.
Treatment protocols are built from that analysis. IV vitamin therapy for someone with oxidative stress markers and low CoQ10 looks different from IV vitamin therapy for someone with B vitamin deficiency and elevated homocysteine — the same delivery method, but different formulations. Peptide protocols for someone with low IGF-1 and poor sleep architecture differ from peptide protocols for someone with adequate GH output but elevated inflammatory markers, accelerating cellular senescence.
The LIVV Cardiff members-only longevity club includes twice-yearly comprehensive biomarker panels as part of the membership, along with ongoing protocol management based on what the data shows at each testing interval. Full details are on the LIVV Cardiff membership page. For people not in the membership, LIVV Little Italy (800 West Ivy St, Suite A, San Diego, Mon-Fri 9 am–5 pm) and LIVV Cardiff (2027 Newcastle Ave, Cardiff, Tues-Fri 10–6, Sat 10–4, Sun 11–4) both offer standalone comprehensive panels and subsequent consultation to review results and build a protocol.
Frequently Asked Questions
Which biomarkers should I test first if I’ve never done a comprehensive panel?
Start with a panel that covers all five categories — metabolic, inflammatory, hormonal, cognitive, and longevity — rather than isolating one area. The value of comprehensive testing comes from seeing the full picture at once: a person might have normal CRP but severely elevated insulin resistance, or normal thyroid TSH but abnormal Free T3 that explains their symptoms. Testing only the markers that match your chief complaint misses the connective tissue between systems. A full baseline panel at LIVV covers all of these from a single blood draw and provides the foundation for building any subsequent protocol.
How is hs-CRP different from standard CRP, and which should I test?
Standard CRP testing detects CRP above 10 mg/L — useful for identifying acute infection or injury, but too crude for cardiovascular risk assessment. High-sensitivity CRP (hs-CRP) detects CRP down to 0.1 mg/L, which is the range relevant to chronic low-grade inflammation and cardiovascular risk prediction. For preventive health purposes, hs-CRP is the correct test. Standard CRP is a clinical tool for acute illness, not a preventive medicine marker. Most comprehensive health panels automatically use the high-sensitivity assay.
Can biomarker testing tell me if I have a hormone imbalance before I have symptoms?
Yes — and this is one of the most practically useful applications of regular testing. Hormonal imbalances develop gradually, and the early stages are subclinical: the numbers are moving in an unfavorable direction but haven’t yet crossed diagnostic thresholds or produced symptoms distinct enough to prompt concern. Free testosterone, for example, can be low-normal for years before the effects on body composition, mood, and energy are obvious enough to connect to a hormonal cause. Testing before symptoms appear gives a head start on intervention.
How soon after starting a protocol should I retest to see if it’s working?
It depends on the marker and the intervention. Inflammation markers like CRP respond relatively quickly to dietary changes, sleep improvements, or anti-inflammatory protocols — 8 to 12 weeks is a reasonable retesting interval for these. Hormone levels stabilize within 4 to 8 weeks of an optimization protocol, making that the typical first retest window for TRT or thyroid optimization. Markers that reflect cumulative cellular aging — telomere length, epigenetic age, AGEs — change slowly, and 6 to 12 months is the appropriate interval for meaningful change to accumulate. The general principle: retest frequently enough that you catch interventions that aren’t working, infrequently enough that change has had time to occur.
What’s the difference between a standard blood panel from my doctor and a comprehensive panel at LIVV?
A standard annual physical typically includes a CBC, basic metabolic panel, fasting lipids, and TSH — about 10 to 15 markers aimed primarily at identifying existing disease. A comprehensive LIVV panel adds fasting insulin, HOMA-IR, hs-CRP, IL-6, homocysteine, complete thyroid function (including Free T3, Reverse T3, and antibodies), sex hormone panel (total and free testosterone, estradiol, progesterone, DHEA-S, SHBG), IGF-1, BDNF, vitamin D, B12, magnesium, CoQ10, and longevity markers including telomere length and oxidative stress indicators. The standard panel is designed to catch diagnosable diseases. The comprehensive panel is designed to identify what’s driving your biological trajectory — which is a different question with different clinical utility.
Schedule Comprehensive Biomarker Testing with LIVV
If you want to move from guesswork to data in managing your health, comprehensive biomarker testing at LIVV Natural gives you the full biological picture in a single appointment. The testing is interpreted by the LIVV medical team, and findings are translated directly into a treatment protocol tailored to your specific results — not a standard panel with standard recommendations. A free 15-minute introductory call is available if you want to discuss which panel is most appropriate before booking. Book through the LIVV appointments page or contact LIVV directly at 760-454-4600.