Blog
Biological Age vs. Chronological Age: What the Number on Your Birth Certificate Doesn’t Tell You

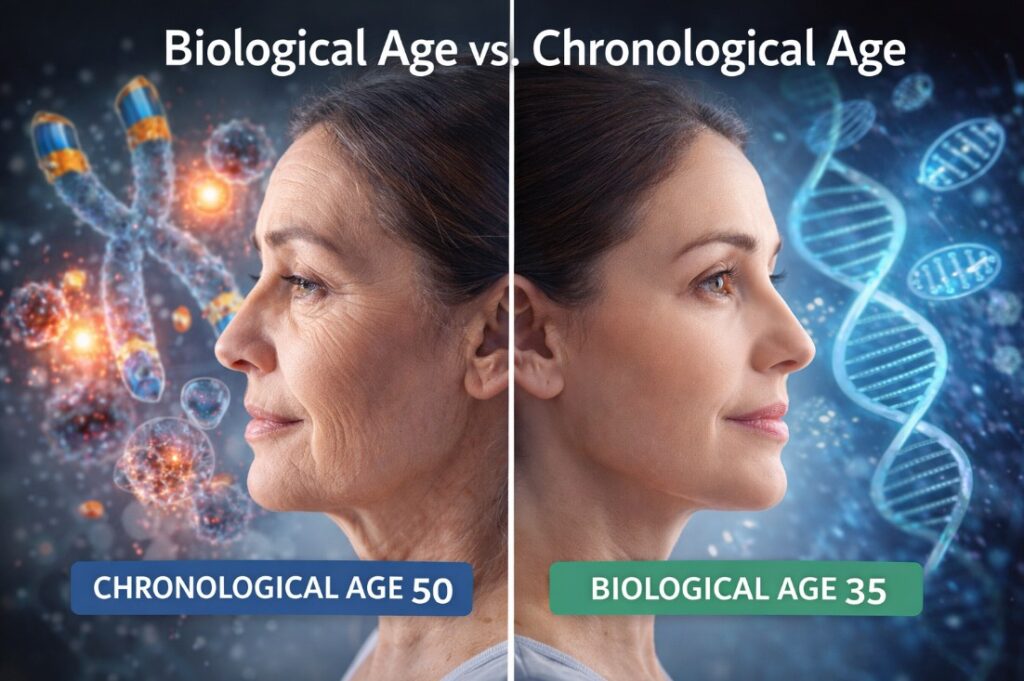
Why Biological Age vs. Chronological Age Is the Right Question
Your chronological age is fixed — it increases one year at a time, regardless of what you do. Your biological age is not. It reflects what’s actually happening in your cells: how well your mitochondria are producing energy, how long your telomeres are, how much inflammation is running at baseline, and what pattern of gene expression your DNA is currently following. Two people who are both 50 chronologically can have biological ages that differ by 15 years or more, and that gap shows up in everything from cardiovascular risk to cognitive performance to how quickly they recover from illness.
This distinction matters because most standard healthcare — annual physicals, routine blood panels, symptom-based medicine — is organized around chronological age. It treats a 55-year-old as though all 55-year-olds are in the same biological situation, which they clearly aren’t. Precision longevity medicine starts from a different premise: that your biological age is measurable, that the gap between biological and chronological age is a clinically actionable number, and that the right protocols can move it.
Genetics accounts for roughly 20 to 25% of how long the average person lives. The other 75 to 80% is determined by lifestyle and environment — diet, sleep, stress, physical activity, and the quality of the interventions applied to cellular aging over time. This isn’t an optimistic estimate from a wellness company. It comes from the Danish Twin Study, one of the largest longitudinal studies of heritability and longevity ever conducted, and it’s been reproduced consistently across subsequent research.
What Biological Age Actually Measures
Chronological age counts years. Biological age counts cellular wear. It’s an estimate of how far along the aging process your cells actually are — based on the accumulation of epigenetic changes, the state of your telomeres, the efficiency of your mitochondria, your inflammatory load, and other markers of cellular health that collectively determine how well your body is functioning relative to where it should be.
The concept matters because aging is not a uniform process. Some tissues age faster than others in the same person. Some people’s cells age faster than their chronological peers because of chronic stress, poor sleep, high inflammatory load, or long-term exposure to environmental toxins. Others age more slowly because of genetic advantages, consistent lifestyle habits, or targeted interventions. Biological age captures this variation in a way that a date of birth cannot.
It’s also worth understanding what biological age is not. It isn’t a death sentence or a guarantee. A high biological age relative to chronological age indicates elevated cellular stress and a higher statistical risk of age-related disease — but it’s a modifiable number, not a fixed one. The same measurements that reveal a gap between biological and chronological age also tell you which pathways are driving it, which points directly to what interventions are most likely to close that gap.
How Biological Age Is Measured: The Four Main Methods
DNA Methylation (Epigenetic Clocks)
Epigenetic clocks are the current gold standard for biological age measurement. They work by analyzing DNA methylation patterns — chemical tags that accumulate on the genome over time and regulate gene expression without changing the underlying DNA sequence. The Horvath clock, published in Genome Biology in 2013, was trained on over 8,000 samples across 51 tissue types and achieved a 97% correlation between predicted biological age and actual chronological age. What made it scientifically useful was not just accuracy, but the fact that deviations from predicted age — being biologically older or younger than your calendar age — correlate with mortality risk and disease outcomes independent of other risk factors.
Since Horvath’s original clock, several generations of more refined estimators have been developed. Hannum’s clock uses blood-specific methylation sites. PhenoAge and GrimAge incorporate biological markers alongside methylation patterns and are particularly predictive of all-cause mortality and disease onset. Each clock measures a slightly different dimension of the aging process, and in clinical practice, using multiple clocks provides a more complete picture of where biological aging is occurring and at what rate.
The practical implication is significant: epigenetic clocks can reveal that your blood is aging at one rate, your brain at another, and your skin at a third — reflecting differences in cumulative stress, lifestyle, and cellular maintenance across tissues. This level of resolution makes epigenetic testing the most informative single measurement currently available for biological age assessment.
Telomere Length Analysis
Telomeres are the protective sequences at the ends of chromosomes that shorten with each cell division. When they reach a critically short length, cells enter senescence — a state in which they stop dividing, stop contributing productively to tissue function, and begin secreting inflammatory signals that degrade surrounding cells. Telomere length is therefore a direct measure of cellular replicative history and remaining capacity.
Average telomere length declines about 20 to 40 base pairs per year in most adults, but this rate is heavily influenced by lifestyle: chronic psychological stress, poor sleep, a high-inflammatory diet, and smoking all accelerate shortening, while regular exercise, adequate sleep, and anti-inflammatory nutrition slow it. A prospective trial published in PMC found that 60 sessions of hyperbaric oxygen therapy produced over 20% increases in telomere length across multiple immune cell types in healthy adults over 64 — a magnitude of change that substantially exceeds what lifestyle interventions alone have produced in comparable timeframes. Senescent cell percentages decreased by up to 37% in the same subjects.
Telomere length testing is typically done from a blood sample and reported as an average length in kilobases, often compared against age-matched reference populations. It provides a useful complement to epigenetic clock data — methylation clocks capture the epigenetic changes that accompany aging, while telomere length captures the cumulative mechanical aging of cells through division.
Mitochondrial Function Assessment
Mitochondria are not just cellular energy producers — they’re central regulators of the entire cellular aging process. They generate ATP through oxidative phosphorylation, regulate apoptosis, produce reactive oxygen species as a byproduct of normal function, and respond dynamically to metabolic signals. When mitochondrial function declines, the downstream effects are broad: lower cellular energy output, higher oxidative stress, impaired DNA repair, and a shift toward senescence in affected cells. The detailed mechanisms behind this are covered in the mitochondrial health and aging overview on the LIVV blog.
Clinically, mitochondrial function is assessed through a combination of indirect markers — lactate-to-pyruvate ratios, organic acid profiles, ATP production rates in peripheral blood cells, and CoQ10 levels — and direct measures of cellular energy metabolism. This type of testing identifies mitochondrial decline before it produces obvious symptoms like fatigue or exercise intolerance, which is precisely when it’s most addressable.
Inflammatory Markers and Metabolic Biomarkers
Chronic low-grade inflammation — sometimes called inflammaging — is one of the most consistent predictors of accelerated biological aging and age-related disease. CRP, IL-6, TNF-alpha, and homocysteine all correlate with biological age acceleration and with a higher risk of cardiovascular disease, cognitive decline, and cancer. These markers are standard in comprehensive blood panels and provide an actionable picture of where inflammation is being driven from — whether by visceral fat, gut dysbiosis, hormonal imbalance, or other upstream factors.
Advanced glycation end products (AGEs) are another aging-relevant metabolic marker. They form when glucose molecules bind to proteins or lipids in a non-enzymatic reaction, accumulating in tissue over time and contributing to arterial stiffness, kidney dysfunction, and skin aging. A foundational biomarker panel that covers inflammatory markers, metabolic indicators, hormone levels, and oxidative stress provides the baseline data needed to identify which pathways are driving biological age acceleration in a specific individual.
Lifestyle Factors That Drive Biological Age
Diet and Nutritional Status
Dietary patterns affect biological age through multiple pathways simultaneously. Anti-inflammatory diets — high in polyphenols, omega-3 fatty acids, antioxidants, and fiber — reduce CRP and IL-6, support the gut microbiome diversity that affects epigenetic maintenance, and provide the methyl donors (folate, B12, choline) that support DNA methylation fidelity. Diets high in refined carbohydrates, processed seed oils, and ultra-processed food drive the opposite: elevated inflammatory markers, glycation, oxidative stress, and measurable acceleration of epigenetic aging clocks.
Intermittent fasting and time-restricted eating produce effects on biological aging that go beyond caloric intake. Fasting periods trigger autophagy — the cellular cleanup process that clears damaged proteins and dysfunctional organelles. They also stimulate mitochondrial biogenesis through AMPK and SIRT1 activation, both of which are NAD+-dependent processes. The net effect of consistent time-restricted eating is a measurably lower inflammatory burden and better mitochondrial function, both of which reduce biological age.
Sleep and Circadian Alignment
Sleep is the body’s primary biological maintenance window. Growth hormone is predominantly secreted during the first two hours of slow-wave sleep, driving tissue repair and protein synthesis. Cellular waste clearance through the glymphatic system is most active during deep sleep. DNA repair activity peaks overnight. Melatonin, secreted by the pineal gland in response to darkness, coordinates circadian gene expression throughout the body, including in skin, liver, and immune tissue. Chronic sleep disruption — whether through short duration, late timing, or poor quality — bypasses all of these processes and produces measurable increases in inflammatory markers, cortisol dysregulation, and accelerated epigenetic aging.
Circadian misalignment — working night shifts, irregular sleep schedules, or habitual late-night light exposure — affects biological age independently of sleep duration. The circadian clock regulates gene expression in almost every cell type, and when it’s chronically disrupted, the downstream effects on cellular maintenance are widespread and cumulative.
Chronic Stress and Cortisol
Psychological stress accelerates biological aging through the HPA axis: stress activates cortisol secretion, which, in sustained amounts, suppresses immune function, promotes visceral fat accumulation, increases systemic inflammation, and directly shortens telomeres. The mechanism involves glucocorticoid receptor signaling in immune cells, which reduces telomerase activity — exactly the enzyme responsible for maintaining telomere length. People with chronically elevated cortisol consistently show older biological ages on telomere length assays and epigenetic clocks, even when other lifestyle factors are controlled for.
Stress-reduction interventions that measurably reduce cortisol — regular meditation, exercise, social connection, nature exposure — produce real effects on biological aging markers over time. This is not a soft claim about wellness. It’s documented in prospective studies that track epigenetic aging before and after stress interventions, and the changes are in the same direction as those produced by more direct clinical interventions.
Exercise
Exercise produces some of the most well-documented benefits for biological age of any modifiable lifestyle factor. High-intensity interval training stimulates mitochondrial biogenesis — the creation of new mitochondria — through PGC-1α activation. Resistance training preserves muscle mass, which is one of the strongest predictors of longevity independent of other variables. Zone 2 aerobic training (moderate sustained effort) optimizes mitochondrial efficiency and substrate utilization. Regular exercise consistently reduces epigenetic age acceleration, maintains telomere length, and lowers inflammatory markers.
The combination matters more than the intensity. HIIT a few times per week alongside regular resistance training and daily moderate movement produces broader biological age benefits than any single modality alone. Sedentary behavior — defined as extended periods of sitting regardless of exercise frequency — independently accelerates biological aging, which means that high daily step counts and movement throughout the day matter alongside structured exercise.
What Accelerates Biological Aging
Beyond lifestyle, several biological processes directly drive biological age when they run unchecked.
Chronic inflammation (inflammaging): Low-grade, persistent inflammation is both a cause and consequence of biological aging. It accelerates telomere shortening, promotes cellular senescence, impairs mitochondrial function, and creates a tissue environment hostile to cellular repair. The drivers of chronic inflammation include visceral adiposity, gut dysbiosis, unresolved psychological stress, poor sleep, hormonal imbalances, and environmental toxin exposure.
Mitochondrial dysfunction and oxidative stress: As mitochondria age, their efficiency declines, and they produce more reactive oxygen species (ROS) relative to ATP output. Excess ROS damages DNA, proteins, and lipids — the oxidative damage that accumulates in aging tissue. Mitochondria with damaged DNA or dysfunctional membranes are less efficiently cleared through mitophagy (targeted cellular cleanup), allowing a growing population of dysfunctional mitochondria to coexist with healthy ones and drag down overall cellular energy production.
Cellular senescence: Senescent cells are cells that have exited the cell cycle and lost productive function but haven’t yet been cleared. They accumulate with age and secrete a cocktail of inflammatory proteins, matrix-degrading enzymes, and growth factors that accelerate aging in surrounding tissue — a process called the senescence-associated secretory phenotype (SASP). The accumulation of senescent cells is now considered a primary driver of tissue aging, and interventions that reduce senescent cell load (senolytics) are one of the most active areas in longevity research.
Advanced glycation end products (AGEs): Glycation is the non-enzymatic bonding of glucose to proteins and lipids, forming AGEs that accumulate in long-lived proteins like collagen, elastin, and lens crystallins. AGEs stiffen arteries, impair kidney function, contribute to neurodegeneration, and are directly measured in skin autofluorescence testing. High-sugar diets, poor glycemic control, and excessive cooking temperatures all increase AGE accumulation rates.
How to Slow and Reverse Biological Aging: Clinical Interventions
NAD+ Therapy: Restoring Cellular Energy and DNA Repair Capacity
NAD+ is a cofactor required by two of the most important enzyme families in cellular aging: sirtuins (which regulate DNA repair, gene expression, and mitochondrial biogenesis) and PARPs (which coordinate DNA damage repair). NAD+ levels decline by 40 to 50% between young adulthood and middle age across most tissues, which impairs both of these systems simultaneously. The connection between NAD+ decline and the hallmarks of aging is one of the most well-established mechanisms in longevity biology. NAD+ IV therapy delivers NAD+ directly into the bloodstream, producing a faster and larger increase in tissue levels than oral precursors like NMN or NR — a relevant distinction for people whose baseline levels are already significantly depleted.
NAD+ restoration supports multiple biological aging pathways at once: sirtuin activation promotes mitochondrial biogenesis and collagen maintenance, PARP activity clears accumulated DNA damage, and SIRT1-mediated autophagy removes senescent proteins and dysfunctional organelles. This multi-pathway effect is why NAD+ features in most serious longevity protocols — it’s not addressing one specific mechanism but rather restoring the substrate that enables multiple cellular maintenance systems to function.
Peptide Therapy: Telomere Support and Cellular Regeneration
Several peptides have documented effects on biological aging mechanisms. Epithalon, a synthetic tetrapeptide derived from pineal gland extracts, activates telomerase and has been shown in cell culture studies to extend telomere length in human somatic cells through upregulation of hTERT. Growth hormone secretagogues like CJC-1295/Ipamorelin and Tesamorelin increase IGF-1 and stimulate cellular repair and protein synthesis across multiple tissues. MOTS-c is a mitochondrial-derived peptide that activates AMPK, reduces cellular stress, and improves metabolic function — one of the few peptides that acts directly on mitochondrial signaling pathways. The full breakdown of longevity and anti-aging peptides covers the mechanism and evidence for each of these in more detail.
Hyperbaric Oxygen Therapy (HBOT)
HBOT delivers 100% oxygen at elevated atmospheric pressure, dissolving it directly into blood plasma at concentrations not achievable through normal breathing. Beyond its established uses in wound healing and tissue repair, HBOT has documented effects on cellular aging. A prospective trial on HBOT and telomere length enrolled 35 healthy adults aged 64 and older for 60 daily HBOT sessions. Telomere length in multiple immune cell types increased by more than 20%, with B cells showing increases of up to 37.6%. Senescent T-helper cell percentages decreased by 37.3% post-HBOT. No other currently available intervention had produced comparable telomere elongation in a comparable timeframe at the point of publication — the researchers noted that HBOT’s effect exceeded what had been reported from lifestyle modification and exercise studies.
The mechanism behind HBOT’s aging effects involves the hyperoxic-hypoxic paradox: repeated cycles of high oxygen exposure followed by return to normal oxygen levels trigger HIF-1α expression, which activates regenerative pathways that normally engage during hypoxic stress. This includes mitochondrial biogenesis, angiogenesis, and senolytic activity. The HBOT anti-aging overview on the LIVV blog covers the full scope of clinical data on HBOT for aging applications.
Glutathione and IV Antioxidant Therapy
Oxidative stress is both a driver of biological aging and a consequence of it. Glutathione — the body’s primary intracellular antioxidant — declines with age, leaving cells progressively less capable of neutralizing the ROS produced by normal mitochondrial function and external environmental exposures. Glutathione IV therapy restores glutathione to functional levels more rapidly than oral supplementation (which has poor bioavailability in most forms), reducing the oxidative burden on cells and supporting the DNA repair and cellular maintenance systems that depend on a low-oxidative environment to function.
Contrast Therapy: Cold and Heat for Mitochondrial and Cardiovascular Function
Alternating cold plunge and infrared sauna sessions — contrast therapy — produces compounding effects on biological aging markers. Sauna exposure at high temperatures mimics the effects of sustained exercise on cardiovascular function and has 20-year cohort data associating frequent sauna use with dramatically lower cardiac mortality. Cold exposure activates brown adipose tissue, stimulates norepinephrine release, and triggers mitochondrial biogenesis through the same cold-shock protein pathways activated by exercise in cold conditions. Together, these thermal stressors produce mitochondrial and cardiovascular adaptations that address two of the primary mechanisms of biological age acceleration.
Can Lifestyle Interventions Measurably Reduce Biological Age?
The answer from clinical research is yes — with specific protocols and measurable endpoints. A randomized controlled trial published in PMC (Fitzgerald et al., 2021) enrolled 43 healthy adult males aged 50 to 72 in an 8-week program combining dietary guidance (emphasizing methylation-supportive foods), sleep optimization, exercise protocols, relaxation practices, and targeted supplementation. Biological age was measured using the Horvath DNAmAge clock from saliva samples. At the end of the 8 weeks, the treatment group had biological ages averaging 3.23 years younger than the control group (p=0.018). Compared to their own baseline, treatment group participants were 1.96 years biologically younger, while the control group aged 1.27 years over the same 8-week period.
This study demonstrates several things simultaneously: that biological age as measured by epigenetic clocks is responsive to lifestyle intervention over a short timeframe, that the rate of biological aging can be meaningfully slowed or reversed without pharmaceutical intervention, and that the specific components of the intervention — diet, sleep, stress management, and exercise — each likely contribute independently to the effect. It also establishes that the gap between biological and chronological age is not fixed — it’s a variable that responds to the inputs you provide your cells.
LIVV Cardiff’s Approach to Biological Age Optimization
LIVV Cardiff’s longevity protocols start from the same premise as the research: biological age is measurable, it reflects real differences in cellular health and disease risk, and it’s modifiable with the right inputs. Every Cardiff membership begins with comprehensive biomarker testing that covers epigenetic age markers, inflammatory burden (CRP, IL-6, TNF-alpha), metabolic indicators, hormone panels, oxidative stress markers, and mitochondrial function assessments. This establishes a biological age baseline specific to the individual — not an assumption based on calendar years.
From that baseline, the LIVV medical team develops a treatment plan that addresses the specific pathways driving biological age acceleration in that person. For someone with elevated inflammatory markers and mitochondrial dysfunction, the priorities look different than for someone whose primary issue is hormonal decline and low NAD+ levels. The LIVV Cardiff ethos describes the broader philosophy behind this precision medicine approach — the point is that a 55-year-old with a biological age of 48 and one with a biological age of 63 need different protocols, and the data is the starting point for determining which is which.
Available interventions at LIVV Cardiff include NAD+ IV therapy, peptide therapy (including Epithalon, MOTS-c, GH secretagogues, and others selected based on lab data), HBOT sessions, glutathione and IV vitamin therapy, hormone optimization for both men and women, contrast therapy, and lifestyle medicine consultations covering nutrition, sleep, exercise, and stress management. The protocols are updated at regular intervals based on follow-up testing, tracking whether biological age markers are moving in the expected direction.
For people interested in understanding their biological age baseline and what protocols are most appropriate for their specific profile, the latest anti-aging protocols overview on the LIVV blog covers the current state of the field. The Cardiff membership provides the clinical infrastructure — testing, medical team access, and ongoing protocol management — to apply those protocols systematically over time rather than trying single interventions in isolation.
Frequently Asked Questions
How accurate are biological age tests?
The accuracy depends on which test you’re using and what ‘accuracy’ means in context. Epigenetic clocks based on DNA methylation — the Horvath clock, PhenoAge, GrimAge — were developed against datasets of thousands of samples and achieve correlations with chronological age above 95% when used to estimate age in an unknown sample. But for individuals, the clinically relevant question isn’t just ‘how closely does this match my birth year’ — it’s ‘does a deviation between my biological and chronological age predict health outcomes?’ The answer there is also yes: epigenetic age acceleration (being biologically older than your calendar age) is associated with higher all-cause mortality, cardiovascular risk, and cognitive decline risk independent of other factors. The test’s value is in the deviation from prediction, not the prediction itself.
What is a good biological age relative to chronological age?
The goal for most people is to have a biological age that is equal to or lower than their chronological age. The research on Blue Zones populations — the world’s longest-lived communities — consistently shows that centenarians have biological ages well below their calendar ages, driven by sustained lifestyle factors rather than exceptional genetics. For most adults, even closing a 5 to 10 year gap between biological and chronological age represents a substantial reduction in the risk of age-related disease and a meaningful improvement in cellular function. What constitutes ‘good’ also depends on which markers are being measured — someone can have an excellent epigenetic clock result while still carrying elevated inflammatory markers that require attention.
Can biological age be reversed, not just slowed?
Yes — with the caveat that the research on actual reversal (biological age going lower than the starting point) is relatively recent, and the effect sizes, while real, are not unlimited. The Fitzgerald et al. RCT showed participants averaging 1.96 years biologically younger after 8 weeks of lifestyle intervention. The HBOT telomere study showed 20 to 38% increases in telomere length over 3 months. Epithalon has cell-culture evidence for telomere extension through telomerase activation. These are not speculative — they are documented in peer-reviewed research. But they are also not a guarantee of dramatic age reversal for every individual in every situation. The most accurate framing is that certain interventions, applied consistently, can measurably reduce biological age over months to years — and that the effects are additive when protocols address multiple aging pathways simultaneously.
How often should I test my biological age?
Twice a year is the standard frequency recommended in most precision longevity protocols — sufficient to capture meaningful change without over-testing. A single measurement establishes a baseline; the second measurement, 6 months later, shows whether the protocols in use are moving the numbers. Epigenetic clock results from saliva or blood samples don’t change week-to-week the way, say, blood glucose does — changes accumulate over months, which is the appropriate window for assessment. Inflammatory markers and metabolic indicators can be retested more frequently (quarterly) since they respond faster to interventions and provide useful feedback on whether the protocol is working before the next epigenetic assessment.
What’s the single most impactful thing I can do to reduce my biological age?
There isn’t one universal answer because biological age acceleration is driven by different factors in different people, which is precisely why testing matters. For someone with severely disrupted sleep, fixing sleep quality produces faster and larger effects on inflammatory markers and epigenetic aging than almost any clinical intervention. For someone with adequate sleep but chronic psychological stress and elevated cortisol, stress management produces the biggest return. For someone with measurably low NAD+ levels, restoration through IV therapy addresses a bottleneck that other interventions can’t compensate for. The honest answer is: test first to find out what’s driving your biological age acceleration, then address that specific pathway as the primary intervention. Everything else should be secondary to that.
Measure Your Biological Age and Build a Protocol Around It
If you want to know your actual biological age rather than making assumptions based on how you feel, LIVV Cardiff and LIVV Little Italy both offer comprehensive biomarker testing that covers the full range of biological aging markers — epigenetic indicators, inflammatory burden, mitochondrial function, hormones, and metabolic health. The results give you a specific biological age estimate and identify which pathways need the most attention.
For people who want ongoing protocol management alongside the testing — regular follow-up, protocol adjustments based on data, and access to the full range of clinical interventions — the LIVV Cardiff membership provides that structure. Membership includes twice-yearly comprehensive biomarker panels, access to NAD+ IV therapy, HBOT, peptide protocols, contrast therapy, and regular consultations with the LIVV medical team.